Type 2 diabetes (T2DM) is a rapidly evolving global health issue[1] and Asia is the epicenter of this worldwide epidemic.[2] The prevalence of T2DM has been predicted to double from 7.3% in 1990 to 15% in 2050 in Singapore, a multi-ethnic city-state composed of three major ethnic groups (Chinese, Malays and Indians).[3] The ethnic diversity in Singapore provided us a unique opportunity to study the ethnic disparity on T2DM-related adverse outcomes.
Ethnic disparity in the development of diabetic complications, such as cardiovascular disease (CVD) and mortality from CVD, has been reported in many Western countries.[4][5] Such ethnic disparity has also been found among Asians with T2DM in Singapore, showing higher risk for ischemic heart disease and cardiovascular mortality in Indians and Malays compared to Chinese.[6]-[9] In our recent studies in Singapore, we also revealed higher risk for diabetic complications in Indians and Malays compared to Chinese, such as acute myocardial infarction and cardiovascular mortality.[10] [11] Several reasons have been suggested to explain their excess adverse outcomes, including less favorable metabolic and lipid profiles e.g., body mass index (BMI); lifestyle, e.g., less physical activity, less fruit and vegetable consumption; and poorer diabetes control, e.g., higher hemoglobin A1c (HbA1c). Furthermore, we observed higher central arterial stiffness, a well-established biomarker for vasculopathy, and lower level of soluble receptor for advanced glycation endproducts (sRAGE), a molecule with protective function on the vasculature in Malays and Indians than Chinese.[12] However, the mechanisms underlying the ethnic disparity in multifactorial diseases, such as CVD still remain largely unknown.
An inter-arm difference in blood pressure of less than 10 mmHg is classified as normal based on the new clinical guideline for hypertension.[13] Evolving evidence suggested that an inter-arm difference in systolic blood pressure (IADSBP) of 10 mmHg or more is associated with CVD and cardiovascular mortality.[14][15] It is interesting that the prevalence of IADSBP ≥10 mmHg is higher among T2DM patients (~9-10%) when compared to the general population (4.4%).[16][17] Recently, IADSBP ≥10 mmHg has been associated with CVD and cardiovascular mortality in T2DM patients, suggesting that the detection of IADSBP may identify a subpopulation at high risk of vascular events among T2DM patients.[18]
The prevalence of IADSBP ≥10 mmHg varies widely across different studies, depending on ethnicity and background of enrolled individuals.14,15 For example, the prevalence of IADSBP ≥10 mmHg or the mean of IADSBP in Asians[19]-[21] is generally lower than that in Caucasians.[18][22][23] In a multi-ethnic study of atherosclerosis in USA, different prevalence rates of IADSBP ≥15 mmHg were observed in African-Americans, Caucasians and Chinese.[24] However, disaggregated information on IADSBP is limited among Asian sub-ethnicities with T2DM, notwithstanding the existence of clear ethnic disparity in diabetes-related adverse outcomes. Therefore, we aim to investigate ethnic differences in IADSBP in a multi-ethnic T2DM Asian cohort.
METHODOLOGYStudy population and design
The Singapore Study of Macro-angiopathy and Microvascular Reactivity in Type 2 Diabetes (SMART2D) is a cross-sectional study conducted between August 2011 and February 2014 which included a total of 2,057 adults aged 21–90 years with T2DM. Inclusion and exclusion criteria of SMART2D have been previously described.[25] We included 1,233 individuals with bilateral BP measurements in this study. These selected individuals have similar profile compared to the rest of the subjects (detailed data not shown). This study has been approved by our institution’s domain-specific ethics review committee. Individual written informed consent was obtained prior to enrollment in the study.
Clinical and biochemical measurementsBMI was calculated as body weight (kg)/height (m)2. Urinary albumin-to-creatinine ratio (ACR) was determined by urinary creatinine measured by enzymatic method on Roche/Hitachi cobas c system (Roche Diagnostic GmbH, Mannheim, Germany) and albumin measured by a solid-phase competitive chemiluminescent enzymatic immunoassay with a lower detection limit of 2.5 μg/ml (Immulite; DPC, Gwynedd, UK). Estimated glomerular filtration rate (eGFR) was calculated based on a widely used Modified Diet in Renal Disease (MDRD) equation in patients with diabetes.[26] HbA1c was measured based on monoclonal antibody agglutination reaction using a point-of-care immunoassay analyzer (DCA Vantage Analyzer; Siemens, Erlangen, Germany) certified by National Glycohemoglobin Standardization Program. High-density lipoprotein-cholesterol (HDL-C) and low-density lipoprotein-cholesterol (LDL-C) were quantified by enzymatic method using Kodak Ektachem chemistry slides. Total triglyceride was quantified by enzymatic colorimetric method on Roches/Hitachi cobas c system. Carotid-femoral pulse wave velocity (PWV) was measured by the foot-to-foot method using a wellvalidated device, SphygmoCor® (AtCor Medical, Sydney, Australia) as described previously.[27] PWV was expressed as the distance between the two recording sites (meters) divided by transit time (seconds). Total sRAGE was quantified by ELISA (R&D Systems, Minneapolis, MN) according to manufacturer's protocol. The intra- and inter-assay coefficients of variation were 5.7% and 7.7%, respectively. The sensitivity reported by the manufacturer was 4.12 pg/ml.
Blood pressure measurementSequentially repeated measurements of bilateral blood pressures were performed using mercury sphygmomanometers, the “gold standard” device for blood pressure measurement.[28] An appropriately sized cuff is attached to the upper arm, and a series of calibration readings are taken with the mercury sphygmomanometer to ensure that the device is giving accurate readings. After five minutes of seated rest, sphygmomanometer was placed on the arm, alternating right then left, and allowing one minute between each measurement on the same arm. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were calculated from the average of three most consistent readings. At the end of the five-minute rest, heart rate was assessed by an OMRON® digital blood pressure monitor.
Statistical analysisStandard descriptive statistics were used to summarize the characteristics of individuals with T2DM. Normally distributed continuous data were expressed as means and standard deviations (SDs), and skewed variables were expressed as median and inter-quartile range and nature log (ln)-transformed before data analysis. Differences among ethnicity were compared by one-way ANOVA or Kruskal–Wallis test where appropriate.
IADSBP was analyzed as categories (<10 and ≥10mmHg). Univariate logistic regression model was used to evaluate the determinants of IADSBP ≥10 mmHg. Variables that are statistically significant in univariate analysis (p<0.25)[29] (Table S1) or known to potentially affect IADSBP and PWV[15][30]-[32] were added into the model. Age, gender, ethnicity, duration of diabetes, HbA1c, BMI, heart rate, PWV, smoking, hypertension (defined as any SBP ≥140 mmHg or DBP ≥90 mmHg[33]), eGFR, ACR, sRAGE, and commonly used medications in diabetes e.g. angiotensin-convertingenzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs), were added into multivariable logistic regression model. All statistical analyses were performed using IBM SPSS (Version 22). A two-tailed p value of less than 0.05 was considered as statistically significant.
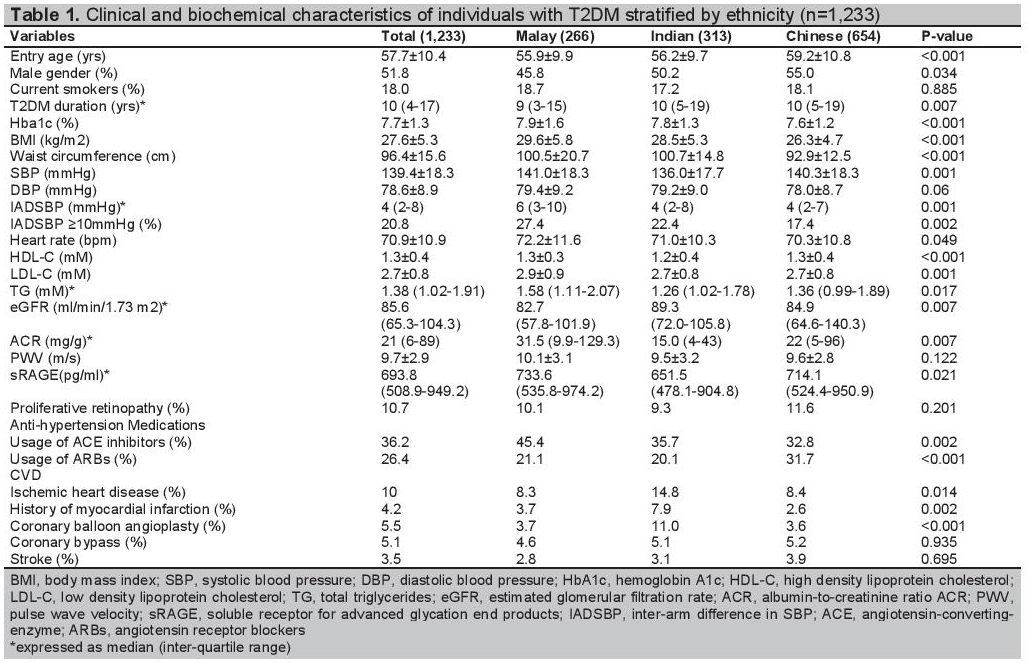
Click here to download Table 1Table 1. Clinical and biochemical characteristics of individuals with T2DM stratified by ethnicity (n=1,233)
In collinearity test for each variable, the tolerance value was >0.2 and variance inflation factor was <3 for all variables, indicating that there was no serious problem with multicollinearity.
Table 1 shows the characteristics of T2DM patients stratified by ethnicity, including 654 Chinese (53.0%), 266 Malays (21.6%) and 313 Indians (25.4%). Compared to Chinese, Malays and Indians were younger and had higher BMI. Malays a had higher heart rate, HbA1c and ACR, and lower eGFR than Chinese and Indians. The percentages of ischemic heart disease, coronary balloon angioplasty, and history of myocardial infarction in Indians are higher than Chinese and Malays. There were no significant differences in percentages of current smoker, coronary bypass operation, stroke and proliferative retinopathy in Malays compared with Indians and Chinese.
Compared to Chinese (5.4±4.8 mmHg), IADSBP is significantly higher in Malays (7.1±6.4 mmHg, p<0.001) and non-significantly higher in Indians (6.1±6.0 mmHg, p=0.132). There is significant higher percentage of IADSBP ≥10 mmHg in Malays (27.4%) and Indians (22.4%) than Chinese (17.4%, P=0.002).
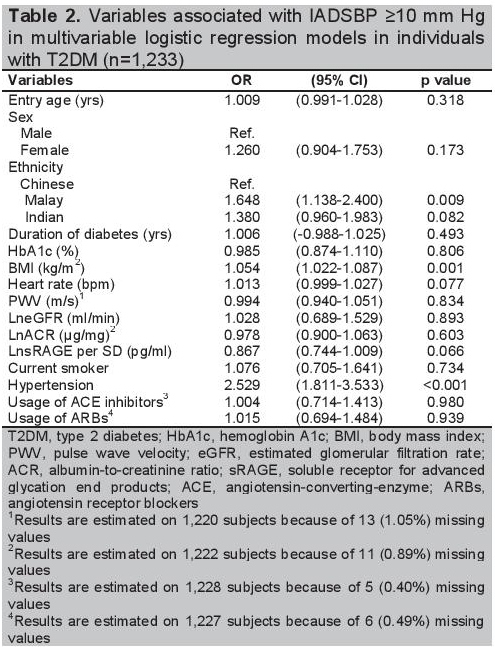
Univariate analysis shows a higher odds for IADSBP ≥10 mmHg in Malays (OR=1.791, 95% CI: 1.279-2.508, p=0.001) than the Chinese and comparable odds of Indians with the Chinese (OR=1.364, 95% CI: 0.997-1.905, p=0.068). Table 2 shows the associations of IADSBP ≥10 mmHg with ethnicity, and clinical and biochemical determinants by multivariable logistic regression models. After adjustment for age, gender, duration of diabetes, hemoglobin A1c, BMI, heart rate, PWV, eGFR, ACR, smoking, hypertension, sRAGE, and usage of hypertension medications, ethnicity remains significantly associated with IADSBP. While odds for Indians are comparable to Chinese (p=0.089), Malays are more likely to have elevated IADSBP of at least 10 mmHg (OR=1.648, 95% CI: 1.138-2.400, p=0.009). BMI (OR=1.054, 95%CI: 1.002-1.087, p=0.001) and hypertension (OR=2.529, 95%CI: 1.811-3.533), p<0.001) are also significant determinants of IADSBP ≥10 mmHg.
Click here to download Table 2Table 2.Variables associated with IADSBP ≥10 mmHg in multivariable logistic regression models in individuals with T2DM (n=1,233)
In the multi-ethnic T2DM Asian cohort, we have, for the first time, demonstrated that while Malays were likely to have IADSBP ≥10 mmHg than the Chinese, the Indians have comparable odds with the Chinese. BMI and hypertension were strongly associated with IADSBP ≥10 mmHg.
The pattern of higher IADSBP in Malays and Indians compared to Chinese in this study is concordant with our previous observation of ethnic-associated greater arterial stiffness[12] and poor T2DM-related outcome.[10][11] Our finding corroborates well with prevailing literature suggesting the existence of a relationship between ethnicity and IADSBP. The IADSBP ≥10 mmHg in Chinese (6.4%, <4 mmHg),[19] Japanese (6.3%, 3.7 mmHg)[20] and Taiwanese (7.1%)[21] are lower than that in Caucasians in UK (8.6%, mean=4.6 mm Hg,[18] 20%, 6.3 mmHg[22]) and USA (9.8%)[23]. In one multiethnic study in USA, different prevalence rates of IADSBP ≥15 mmHg were found in African-Americans (7.4%), Caucasians (5.1%), and Chinese (1.0%).[24] The generally lower IADSBP in Asians than Caucasians might be related to their lower BMI.[20] Consistently, in our study, Chinese had lower BMI, waist circumference and better lipid profile than Malays and Indians. The mechanisms underlying ethnicdisparity in vascular health is complex and may be a composite outcome of non-biological (e.g, social and economic status) and biological factors (e.g., central hemodynamics and arterial stiffness).[34] [35]
We observed that BMI is an independent predictor of IADSBP ≥10 mmHg, which is consistent with previous studies, suggesting a potential causal relationship between obesity and significant IADSBP.[21][30][32] The association between hypertension and IADSBP ≥10 mmHg is in agreement with some reports,[21][30] but not others.[32] The differences in age, SBP, DBP and BP measurement methods may all contribute to the disparate findings. Taken together, weight and BP management may ameliorate IADSBP and hence, CVD in T2DM. The odds for IADSBP ≥10 mmHg were decreased by 13.3% with 1-SD increase in LnsRAGE. Advanced glycation end products (AGEs) play an important role in diabetic complications through interaction with the AGE receptor (RAGE). sRAGE is a multi-ligand receptor on vascular cells that acts as decoy, thus decreasing AGE-cellular binding. Experimental studies have demonstrated the role of AGE-RAGE axis in hypertension.[31] Decreased sRAGE level was found in nondiabetic hypertensive patients.[31] Therefore, further study on sRAGE and IADSBP is desirable.
The usual relative concordance of BP in both arms has been speculated to reflect on-going delicate and finelytuned homeostatic processes. Reasons contributing to the perturbation of this homeostasis include anatomical (e.g., mechanical obstruction secondary to subclavian stenosis) and hemodynamic factors (e.g., endothelial function and atherosclerosis), which could in-part lead to an increase in IADSBP.[32]
CVD is known as a complex disease and can take many forms, such as stroke, myocardial infarction (MI) and IADSBP.[36] ischemic heart disease (IHD). The underlying mechanisms vary depending on the form of the disease. In our study, we found comparable occurrence of IHD, coronary balloon angioplasty and stroke in Malay and Chinese. MI in Malays is higher than Chinese, which agrees with our recent report.[11] It is possible IADSBP is one contributing risk factor for certain forms of CVD, such as MI. In addition to IADSBP, the development of CVD involves many different risk factors, such as genetic factors, hypertension, physical inactivity and unhealthy lifestyle. The measured and unmeasured factors may collectively explain the higher prevalence of IADSBP >10 mmHg in Malays but comparable CVD occurrence compared with Chinese.
The strength of our study is the relatively large sample size of multi-ethnic patients with T2DM living in Singapore. To the best of our knowledge, this is the first study on the association of ethnicity with IADSBP in Asians with T2DM. We are also cognizant of several limitations. We used BP measured sequentially to define IADSBP ≥10 mmHg. This might account for the prevalence of IADSBP ≥10 mmHg (20.8%) higher than most studies in T2DM.[16]-[18] The overestimation of IADSBP could also be attributable to white-coat effects and short-term large BP variability that escaped detection when using sequential measurement method.[14] [15] Therefore, such simple sequential measurement method is recommended for screening in routine practice, while simultaneous measurements should be reserved for confirmation.17 The cross-sectional design of our study precludes any causal-inference between vascular risk factors and IADSBP. Although controlling for potential confounders, we cannot rule out residual confounding from unmeasured factors that could affect blood pressure or IADSBP across three ethnic groups, such as other classes of hypertension medications (i.e., calcium channel blockers) and other renal vascular risk factors (i.e., physical activity, carotid artery intima-media thickness and coronary artery calcium score).[24][37]
In conclusion, our study provides the first evidence that IADSBP ≥10 mmHg in Malays with T2DM may explain their excess adverse diabetes-related vascular outcomes compared to Chinese. Therefore, measuring bilateral BP may be a simple yet clinically useful procedure to identify T2DM patients at high risk for CVD, thereby informing the need to intensify risk factor management.
AcknowledgmentsWe thank Dr. Darren E. J. Seah for his assistance in subject recruitment. We also thank staff from the Singapore Clinical Research Institute (SCRI) for their contribution to study protocol and database design. Study data were collected and managed using REDCap electronic data capture tools hosted at SCRI.
Statement of AuthorshipAll authors have given approval to the final version submitted.
Author DisclosureAll the authors have declared no conflict of interest to the work carried out in this paper.
Funding SourceThis work was supported by Singapore National Medical Research Council Grant PPG/AH(KTPH)/2011. The funder has no role in study design, data collection, analysis, interpretation and manuscript writing.
[1] Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diab Res Clin Pract. 2010;87(1):4-14. DOI.