Glycemic control is at the core of diabetes management. The Diabetes Control and Complications Trial Research Group has shown that strict blood sugar control can reduce the risk of long-term complications.[1] The clinical practice recommendations of the American Diabetes Association for the standards of medical care in diabetes suggest that the glycated hemoglobin (HbA1c) goal for patients in general is <7%, but <6% is preferred if this can be accomplished without significant hypoglycemia.[2] Even if most patients are unsuccessful in bringing their blood sugar levels to near normal values, a 1% reduction in HbA1c level is associated with a relative risk reduction of 21% for any diabetes-related endpoint, 21% for diabetesrelated deaths, 14% for myocardial infarction and 37% for microvascular complications.[3] Because of these findings, great emphasis is placed on controlling blood sugar levels.
Treatment of periodontal disease has been shown to improve glycemic control in diabetic patients. A metaanalysis on the effect of periodontal therapy on glycemic control showed a mean 0.4% reduction in HbA1c in diabetic patients.[4] Data from this meta-analysis mostly came from studies involving patients with type 2 diabetes. Several studies on type 1 diabetics reported significant improvement in periodontal health but not in HbA1c levels after periodontal therapy.[5]-[8] One group, however, found no significant changes even in patients with the best response to periodontal treatment.
Interestingly, Williams and Mahan, long ago reported reductions in insulin requirements following periodontal therapy and tooth extraction in seven of nine patients, with eight of the nine patients having type 1 diabetes.[9] Miller and colleagues, studying a group of nine poorly controlled type 1 diabetics with moderate-to-severe periodontitis, reported a decrease in mean HbA1c level from 8.7 percent pre-treatment to 7.8 percent posttreatment. [10]
Periodontitis is an inflammatory disease initiated by infection. It clinically appears as gingival bleeding and swelling, with an increase in pocket probing depths (PPD). The success of periodontal treatment is usually gauged by the return of tissues to normal appearance and characteristics, e.g., decreased bleeding and reduction in PPD. Recently however, local inflammation observed in periodontal tissues was shown to have a systemic dimension following findings that plasma Creactive protein (CRP) levels are elevated in patients with periodontitis.[11]-[13] CRP is generally regarded as a valuable biomarker of systemic inflammation and cardiovascular disease. Periodontal treatment in type 2 diabetics has shown a weighted mean difference of reductions in CRP after therapy of 0.50 mg/L.[14] More important, CRP is found to be elevated in patients with type 1 diabetes of long duration.[15] The purpose of the current report is to describe the long-term follow-up of a young adult patient with type 1 diabetes managed by intensive oral care and glycemic control measures.
Clinical Relevance Scientific rationale for studyIntensive insulin therapy improves metabolic control in diabetic patients, but does not result in improved clinical periodontal status. Periodontal intervention in type 1 diabetics improves periodontal parameters but not glycosylated hemoglobin levels.
Principal findingThe use of both periodontal intervention and motivation to reinforce diabetes self-management skills resulted in: 46.7% decrease in the fructosamine levels, a net decrease of 1.2% in glycated hemoglobin levels, and a 50% reduction in the C reactive Protein levels
Practical implicationsIntegrating dental care in the comprehensive management of diabetes promotes better glycemic control and compliance among type 1 diabetic patients.
CASEThe patient is a female diagnosed with type 1 diabetes at age 7. She was first examined on March 3, 2012. She injects insulin twice a day, using a combination of isophane and regular insulin. She frequently has to adjust her dose as she experiences hypoglycemia in the morning frequently. The patient owns a glucose meter, but seldom monitored her blood sugar due to the high cost of glucose strips. She went to a dental facility when newly diagnosed, but was apparently turned away because the dentist felt inadequate to treat pediatric diabetic patients. She has not returned for dental care until her visit to this clinic. Scaling and polishing was performed in 3 visits to remove supragingival calcular deposits. Oral hygiene instructions were given and photographs of her teeth were taken (Figure 1). The patient did not return to continue treatment due to lack of money for transportation expenses (Figure 2).
Click here to download Figure 1Figure 1.Patient’s initial appearance, March 3, 2012.

Click here to download Figure 2
Figure 2.After 3 rounds of scaling.

Definitive Treatment
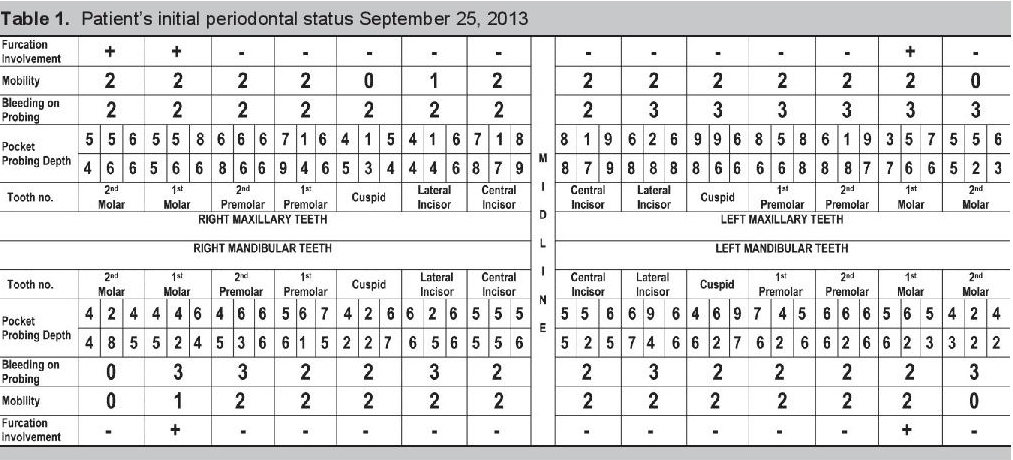
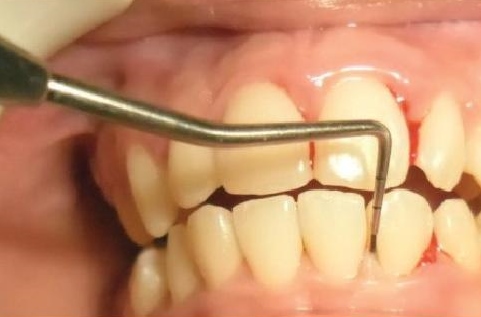
When the patient returned on September 25, 2013, pocket probing depth (PPD) was measured and recorded (Table 1). The PPD is measured in six sites around the tooth, using a periodontal probe with calibrated marking to indicate depth in millimeters (Figure 3). PPD is the distance between the marginal gingiva and the bottom of the gingival sulcus. A pocket probing depth of 4 mm and above is indicative of the presence of periodontitis. Bleeding on probing (BOP) is indicative of the degree of inflammation that is present. A tooth was given a BOP score of: 0 when no bleeding was observed, 1 when a dot of blood appeared on the gingival margin following probing, 2 when a line of blood appeared, and 3 when blood overflowed from the gingival margin (Figure 3). Tooth Mobility is indicative of the amount of alveolar bone loss around the tooth. Mobility was given a score of: 0 when no mobility was detected, 1 when there was less than a millimeter (mm) of mobility detected, 2 when there was a >1 mm mobility detected (Table 1). Caries or tooth decay was found on the following teeth: 15, 26, 27, 36, 46, and 47. Tooth numbers 18 and 28 were impacted. Tooth numbers 13 and 37 had periodontal pockets less than 6 mm while all remaining teeth had pockets greater than 6 mm (Table 1). Except for tooth numbers 47, 46, 36, and 37, all the other teeth had 50 % or less remaining bone support based on the panoramic x-ray (Figure 4).
Click here to download Table 1Table 1.Patient’s initial periodontal status September 25, 2013
Click here to download Figure 3
Figure 3.Example of recording pocket probing depth and bleeding on probing.
Click here to download Figure 4
Figure 4.Patient’s panoramic radiograph September 25, 2013.
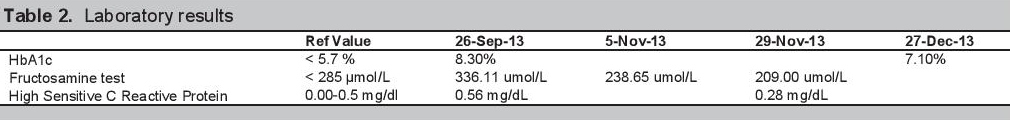
The patient is 151 cm tall and weighs 43.1 kg, with a body mass index of 19 kg/cm2. The following laboratory tests were performed over the course of treatment (see Table 2): glycated hemoglobin assay (HbA1c), fructosamine test, and high sensitive CRP. HbA1c reflects the mean blood glucose concentration over the preceding 1–3 months and has been found in studies to correlate well with the development of diabetes complications.[16]
Click here to download Table 2Table 2.Laboratory results
The fructosamine test is a measure of glycated albumin. It reflects glycemic control over a 2-3 week period. It can be helpful in evaluating the effects of periodontal therapy on glycemic control.[17] Evidence from crosssectional studies have shown that plasma CRP is elevated in periodontitis compared with controls and there is modest evidence on the effect of periodontal therapy in lowering the levels of CRP.[14]
The patient was provided with insulin and glucose strips for blood glucose home monitoring up to November by Colgate Palmolive. The patient was also provided with a toothbrush, triclosan/ copolymer toothpaste, dental floss, an interdental toothbrush, 0.12% chlorhexidine gluconate mouthwash (up to 6 weeks post-root planing) and a 0.075 cetylpridinium chloride mouthwash (after 6 weeks postroot planing). Oral hygiene instructions were again given. The teeth were then divided into sextants and treatment (scaling and root planing) was performed beginning with the anterior teeth. The following teeth were extracted: 16, 15, 26, 27, 32, 31, 41, 42 and 47. During the course of treatment, the patient was given repeat instructions on oral hygiene and diabetes (see Box 1).
Click here to download Box 1Box 1.Diabetes Self-Management Education topics learned in camp reiterated
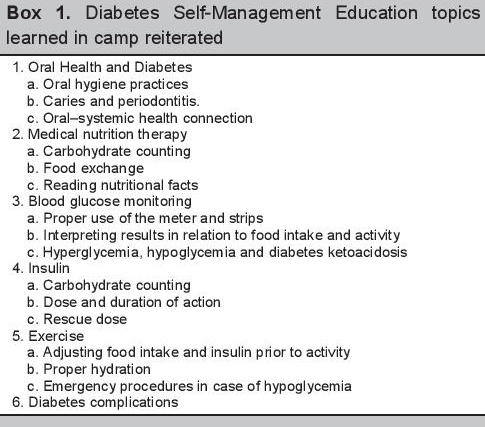
The patient was also asked to keep a food diary, a record of daily blood glucose readings and insulin doses. Additionally, the patient was provided with transportation money and a small allowance.
Overall, the patient’s oral health significantly improved following treatment (Figure 5). Of the patients 28 erupted teeth, 9 (32%) were extracted due to severe mobility, furcation involvement, or pulpal involvement. The patient was unwilling and lacked the resources to return for endodontic treatment. The remaining 19 teeth showed a decrease in PPD measurements [93 (82%) out of 114 sites] and BOP scores [11 (58%) out of 19] (Table 3). Significantly, there was a 50% reduction in the High Sensitive C reactive Protein levels and a 46.7% decrease in the fructosamine assay levels. Initial glycohemoglobin level of 8.3% decreased substantially to 7.1 % (Table 2).
Click here to download Figure 5Figure 5.Marked resolution of inflammation with marked gingival recession.

Click here to download Table 3
Table 3.Periodontal status after root planing

The Institute of Medicine recommends that all health professionals should be educated to deliver patientcentered care as members of an interdisciplinary team, emphasizing evidence-based practice, quality improvement approaches, and informatics.[18] As early as the beginning of the nineteenth century, the call for the integration of medicine and dentistry can be found in the Flexner[19] and Gies[20] Reports. Scientific and technological advances in molecular biology, immunology, and genetics, along with an aging population with more complex health needs, has increasingly linked dentistry and medicine, i.e., the knowledge and skills physicians need related to clinical dentistry and the knowledge and skills dentists need related to clinical medicine are progressively overlapping.[21] Management of oral problems in children with diabetes affords one of the best examples of interprofessional collaboration. The dentist, physician, dietitian and diabetes nurse educator need to coordinate efforts take care of patients effectively.
Summer camps for children with diabetes have the objective of teaching children how to manage their diabetes at home. It encourages children to be independent, self-sufficient, and knowledgeable about their diabetes. It promotes empowerment and at the same time compliance to recommended regimen in oral hygiene, diet, medication, blood sugar monitoring and visits to health professionals. During these camping sessions, interprofessional education occurs among the different health professionals acting as facilitators or counselors during the camp. It allows for sharing of information, strategies, and most of all, it allows health professionals from one discipline to appreciate the role of other health professionals.
Management of periodontal disease for the patient described herein relied heavily on mechanical debridement of crown and root surfaces; still the most reliable way of reducing plaque and calcular deposits. In addition, improved oral hygiene practices (toothbrush and dental floss) and chemotherapeutic agents (cetylpyridinium chloride; chlorhexidine) were employed to prevent recolonization by bacterial pathogens and to reduce gingival inflammation.
An earlier report suggested that type 1 diabetic patients show much improvement in metabolic control after intensive insulin therapy, but no improvement in clinical periodontal status and no changes in the subgingival levels of periodontitis-associated bacteria.[22] Other studies employing periodontal intervention in type 1 diabetics, showed significant improvement in periodontal parameters but not in glycosylated hemoglobin levels.[5]-[8]
What was unique in the management of the patient was the use of both periodontal intervention and motivation to reinforce diabetes self-management skills. Klein and colleagues cited 3 meta-analyses of the effectiveness of Diabetes Self-Management Education in reducing glycated hemoglobin values and reported reductions from 0.32% to 0.76%.[23] The net decrease of 1.2% in glycated hemoglobin levels in this patient, compared to results of previous studies, on type 1 diabetic patients, is considerable. Perhaps the reason for little improvement in glycemic control after periodontal treatment in previous studies was due to the neglect of other factors that affect glycemic control. This suggestion is supported by (i) the improvement in clinical periodontal disease parameters following mechanical cleaning of the teeth, (ii) the initial 30% reduction in fructosamine levels 4 weeks following periodontal therapy, and the 50% reduction in CRP achieved with this patient. However, these results, while encouraging, are based only on a single patient. Thus, it seems reasonable to propose direct testing of this hypothesis in a large interventional study of type 1 diabetics.
Lessons learned in diabetes camps regarding insulin dose adjustment, traditionally taught by physicians, are important tools in curbing rising blood sugar levels. However, most pediatric patients are reluctant to do this at home on their own. Calorie counting and food exchange, taught by dietitians, are effective methods of lowering carbohydrate intake but depends a lot upon the patient’s desire and motivation to follow. Insulin injection and glucose monitoring, taught by diabetes nurse educators, is part of the daily routine of children with diabetes. But, the daily repetitious act becomes monotonous that long term compliance eventually becomes poor. All these are important tools that the diabetic patient can employ to achieve optimum health. The dentist, acting as a diabetes educator, can help improve patient compliance by reinforcing lessons learned in camp during dental visits.
Caries and periodontal disease are the most common oral problems. It is highly preventable, but if left untreated, it can lead to functional problems that can affect dietary needs, psychological problems that can affect self-esteem, and systemic problems that can impair blood glucose control in diabetic patients.
The goal of the dentist is no longer just the improvement of oral health but ultimately the overall health of the patient; and the physician’s goal is to include oral health in the promotion of overall health. Science and patient needs have erased professional boundaries. It is now time to change traditional paradigms regarding oral health and disease so that oral health becomes an integral component of general health. It is now time for the dentist and the physician to share in the responsibility for the oral health and overall health of the patient.
Laboratory tests were sent to the Hi-Precision laboratories. HbA1c was determined using high performance liquid chromatography while fructosamine was determined using a colorimetric reaction with nitrobluetetrasolium. CRP was determined by the agglutination method.
AcknowledgementsI would like to thank the following: the staff of Rainbow Camp, Camp Cope, and Davao Diabetes Camp for inviting me to their camp to share my knowledge in dentistry and learn about diabetes; Dr. AC Dampil and the St. Luke’s Medical Center for providing much needed examination and laboratory tests to rule out any diabetes complications; Mr. Iam Guerrero of Colgate who suggested the idea of making a case report and at the same time helping this particular patient obtain the necessary treatment she needed; Mrs. Liberty Amos Nichols and her daughter Macky Mendoza for donating the glucose strips used by the patient for the duration of the study; and Ms Monica Solis of SciGen and Caesarah Taboada formerly of Novo Nordisk for the insulin used by the patient; Mrs. Becky Fuellos of Children International Philippines, for the insulin syringes, and Dr. Raphael Silva and Dr. Bruce Baum for reviewing the manuscript. Most of all I would like to thank all the children with diabetes who have shared their life and experience with me since 1996.
Ethical ConsiderationPatient consent form has been procured prior to the case report study.
Author Disclosure and Source of FundingThe patient received an honorarium and money from Colgate Palmolive, Philippines for various needs including laboratory tests, panoramic x-ray, and oral hygiene products consisting of toothpastes, toothbrushes, interdental toothbrushes, dental floss, chlorhexidine and cetylpridnium mouthwashes.
The author received an honararium for writing this case report from Colgate Palmolive, Philippines.
[1] Diabetes Control and Complications Trial Research Group (DCCT). The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. New Engl J Med. 1993;329(14):977–86. DOI.